[ad_1]

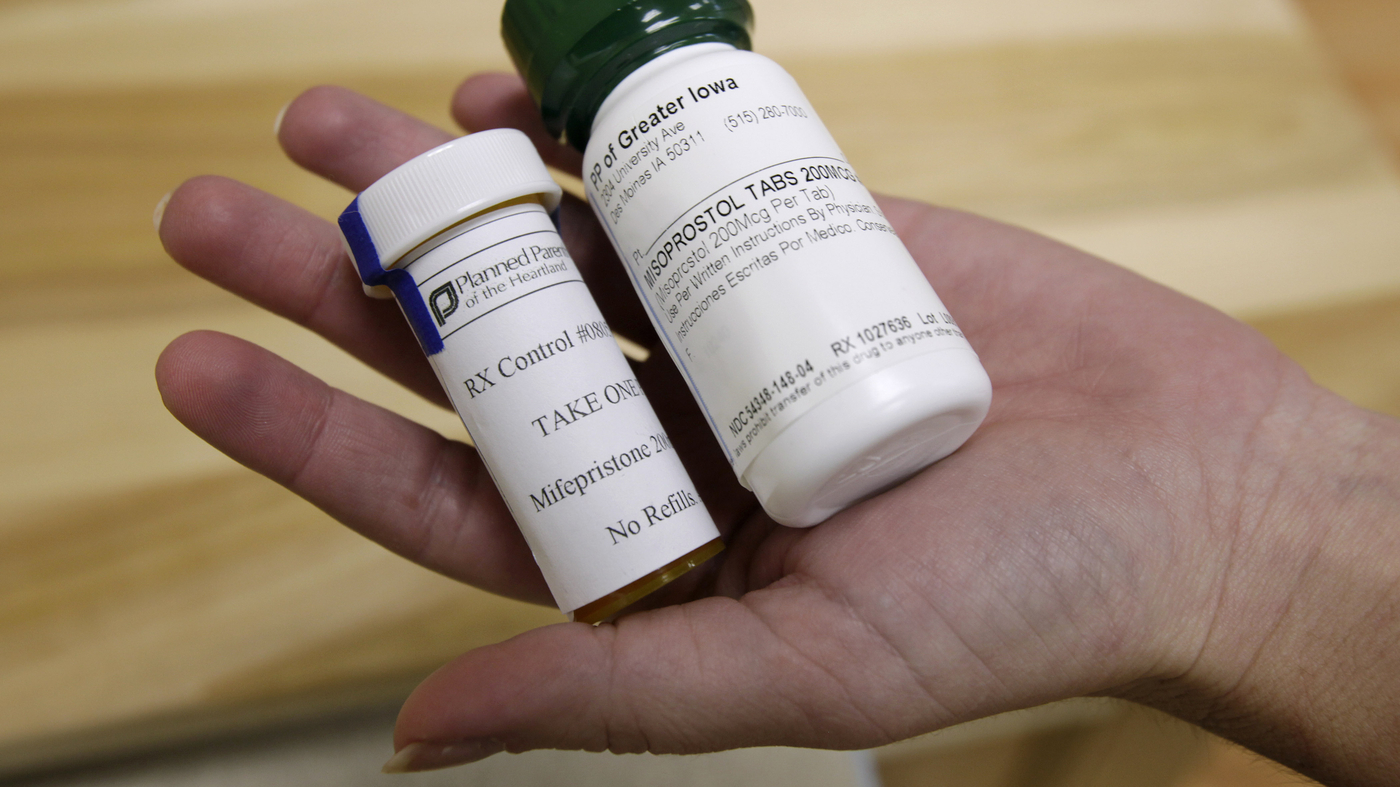
Mifepristone was authorized by the Meals and Drug Administration in 2000 to induce first-trimester abortions together with a second drug, misoprostol.
Charlie Neibergall/AP
conceal caption
toggle caption
Charlie Neibergall/AP

Mifepristone was authorized by the Meals and Drug Administration in 2000 to induce first-trimester abortions together with a second drug, misoprostol.
Charlie Neibergall/AP
A coalition of state attorneys basic is suing the Meals and Drug Administration, accusing the company of excessively regulating the abortion capsule mifepristone.
Mifepristone was authorized in 2000 to induce first-trimester abortions together with a second drug, misoprostol. The lawsuit, filed in federal courtroom in Washington state by a dozen Democratic state attorneys basic, asks the FDA to carry extra layers of regulation above and past these for typical prescribed drugs.
It accuses the FDA “singling out mifepristone…for a novel set of restrictions,” and asks the courtroom to declare the drug to be protected and efficient, and invalidate the extra regulation, generally known as a Threat Analysis and Mitigation Technique.
The go well with comes as a federal choose in a separate case in Texas is contemplating whether or not to overturn the FDA approval of the abortion drug, organising the potential for conflicting rulings by completely different federal judges.
An FDA official says the company doesn’t touch upon ongoing litigation.
[ad_2]